 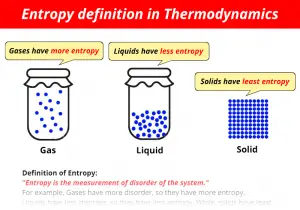
Entropy increases as we go from solid -> liquid -> aqueous -> gas. The second way is to look at the different states of matter between the products and reactants of a chemical equation. This will later be important to do in the next section spontaneous. The first way is to think about each molecu le as ha ving a specific entropy and looking up the value (number) associated that molecule on an entropy table. How can entropy be asked about or displayed in chemistry? The energy is expended by the person when they lift the clothes up from the floor or bed and fold them into the closet or dresser. From Monday to Friday the entropy of the bedroom increases (becomes more messy) but over the weekend what happens to the bedroom? The bedroom gets cleaned by a person putting energy into it. We can also think about the bedroom analogy that we were discussing about before. The energy from the cooking process fuses them together into a larger mass and decreases the entropy of the egg. Just like taking multiple eggs and scrabbling them together and then cooking them. This allows you to make a bigger molecule or a bigger anything. As you put energy into anything (including a molecule) you can decrease its entropy. If you want to know more about these arguments then I sugge st you use this link for an expanded explanation after you read the rest of this section. This is one of the most fundamen tal and misunderstood concepts in science today and is one of the arguments that come up between religious believers and non-religious believers. What is the relationship between entropy and energy? The symbol for the change in entropy is Δ S. Entropy causes larger molecules to break down into smaller molecules over time. We can also think about how entropy affects very small things like molecules. These are all examples of how entropy affects our daily lives. 
Paint starts to become flaky and break off over time. So as time passes a bedroom moves from low entropy (orderly) to high entropy (disorderly). 
By Friday cloths are on your floor, on your bed, and all over (very disorderly). On Monday all your clothes are neatly folded in your dresser or hanging in your closet (very orderly). The easiest example to think of how entropy works is a measure of disorder of your bedroom from Monday to Friday. Something with less entropy (more orderly) has a lower value (number) associated with it. Something with more entropy (more disorderly) has a higher value (number) associated with it. As time goes on the entropy of all things will increase. If ∆S is negative, then the negative signs (from the subtraction and the sign of ∆S) will cancel out, and so as T∆S gets bigger, ∆G will get more positive.It is one of the most confusing scientific t erms, but it is also one of the concepts that people are most familiar with. T is always positive, so if ∆S is positive then a bigger T∆S will make ∆G more negative (since we subtract T∆S). 
As T increases, the T∆S component gets bigger. ∆H is still positive and ∆S is still whatever sign you figured out above. Since ∆H and ∆S don't change significantly with temperature (given in the question), we can assume that they keep the same signs and values: i.e. If ∆G is negative (from the question), is the reaction spontaneous or non-spontaneous?Ģ) Let's use ∆G = ∆H - T∆S again. From these values, we can know for certain whether ∆S is positive or negative (hint: remember that we are subtracting ∆G!).ġ) Knowing the sign of ∆G is enough to say whether the reaction is spontaneous or not under these conditions. Temperature is always positive (in Kelvin). We know (from the question) that ∆G is negative and that ∆H is positive. This looks like a homework question, so I'll give you some hints to get you on the riht path rather than answering directly.ģ) We know that ∆G = ∆H - T∆S. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
